Chemistry
Flesocks class
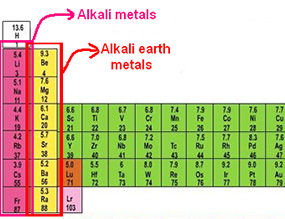
Alkali
Metals
info@mysite.com
Tel: 123-456-7890
Fax: 123-456-7890
By: Rachel De Jesus and Maribel Gonzalez
The Elements in the group:
Lithium (Li)
Sodium (Na)
Potassium (K)
Rubidium (Rb)
Cesium (Cs)
Francium (Fr)
Alkali Metals
Group number: Group 1/group IA
Orbital block: The s block of the periodic table
Family name and its origin : Alkali Metals, the word "alkali" received its name from the Arabic word "al qali," meaning "from ashes". These particular elements were given the name "alkali" because they react with water to form hydroxide ions, creating basic solutions(alkaline solutions).
The Electron configurations of the elements:
Lithium: 1s22s1 or [He] 2s1
Sodium: 1s22s22p63s1 or [Ne] 3s1
Potassium: 1s22s22p63s23p64s1 or [Ar] 4s1
Rubidium: 1s22s22p63s23p64s23d104p65s1 or [Kr] 5s1
Cesium:1s22s22p63s23p64s23d104p65s24d105p66s1 or [Xe] 6s1
Francium:1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p67s1 or [Rn] 7s1
Number of valence electrons: 1
Common ion charges: Alkali metals tend to lose one electron and form ions with a single positive charge. They form ionic compounds in reaction with the halogens (alkali halides).
Group Properties:
-
Highly reactive
-
Soft texture and silvery color
-
Low boiling and melting points
-
Less dense than most elements
-
Low ionization energies
-
Low electronegativities
-
They react quickly with water, producing hydroxides and hydrogen gas
Chemical Reactions:
-
-The alkali metals react with nonmetals in Groups 15 and 16 on the periodic table.
-All the alkali metals react directly with oxygen.
-The alkali metals all react violently with water.
-
-
It's Uses
-
-Lithium is used in batteries, ceramics, and as a neurological drug for mood stabilization.
-
-Sodium is used in medicine, agriculture, food, and to control the amount of ice on the roads during winter months.
-Potassium is used in glass, soap, and to produce fertilizers.
-Rubidium has been used as a component of photocells, to remove traces of oxygen from vacuum tubes and to make special types of glass. Rubidium nitrate is sometimes used in fireworks for a purple color.
-Cesium is used to remove air traces in vacuum tubes.
-Francium has no uses other than basic scientific research because its most stable isotope has a half-life of 22 minutes.

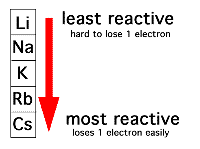
I
Its Sources:
-Lithium was discovered in 1817 by Johann Arfvedson and it is obtained from passing electric charge through melted lithium chloride.
-Sodium was discovered in 1807 by Sir Humphrey Davy obtained from table salts and other foods.
-Potassium was discovered in 1807 by Sir Humphrey Davy obtained from minerals.
-Rubidium was discovered in 1861 by R. Bunsen obtained from lithium production.
-Cesium was discovered in 1860 by Fustov Kirchoff obtained from pollucite, lepidolite.
-Francium was discovered in 1939 by Marguerite Perey obtained from decay of actinium.









(Li)
(Na)
(K)
(Rb)
(Cs)
(Fr)